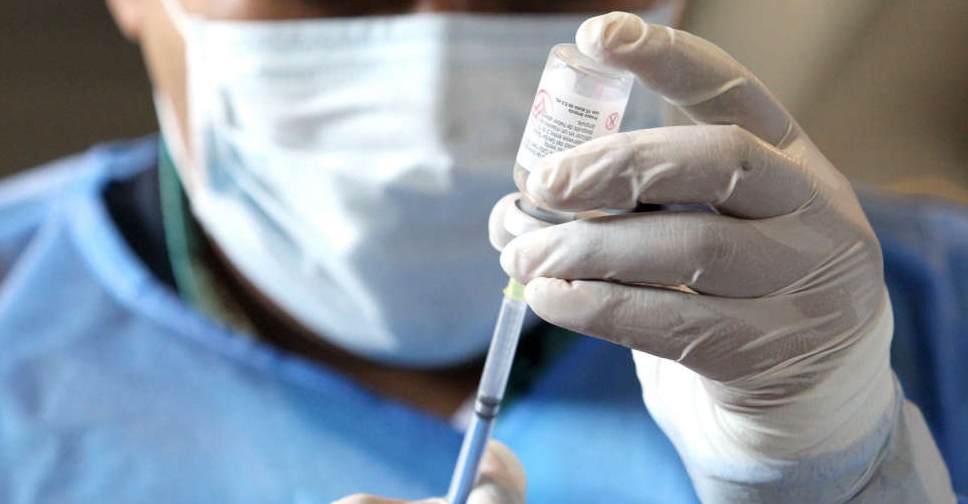
The World Health Organization (WHO) on Thursday issued an emergency use listing for the single-dose COVID-19 vaccine from China-based CanSino Biologics.
The vaccine, Convidecia, is the eleventh shot against the coronavirus to get clearance from the global health agency, whose advisory group recommended its use in people of age 18 years and above.
It was found to have 64 per cent efficacy against symptomatic disease and 92 per cent against severe COVID-19, the agency said.
Other vaccines that have similar clearance from the agency include those made by Pfizer and BioNTech, Moderna, AstraZeneca, Johnson & Johnson and Novavax.




 US hosts Lebanese and Israeli envoys as Israel presses war on Hezbollah
US hosts Lebanese and Israeli envoys as Israel presses war on Hezbollah
 US, Iran may resume talks this week despite port blockade
US, Iran may resume talks this week despite port blockade
 Around 250 missing after Rohingya boat capsizes in Andaman Sea
Around 250 missing after Rohingya boat capsizes in Andaman Sea
 Italy's Meloni suspends defence cooperation deal with Israel
Italy's Meloni suspends defence cooperation deal with Israel
 US-sanctioned tankers pass Strait of Hormuz on first day of US blockade
US-sanctioned tankers pass Strait of Hormuz on first day of US blockade




