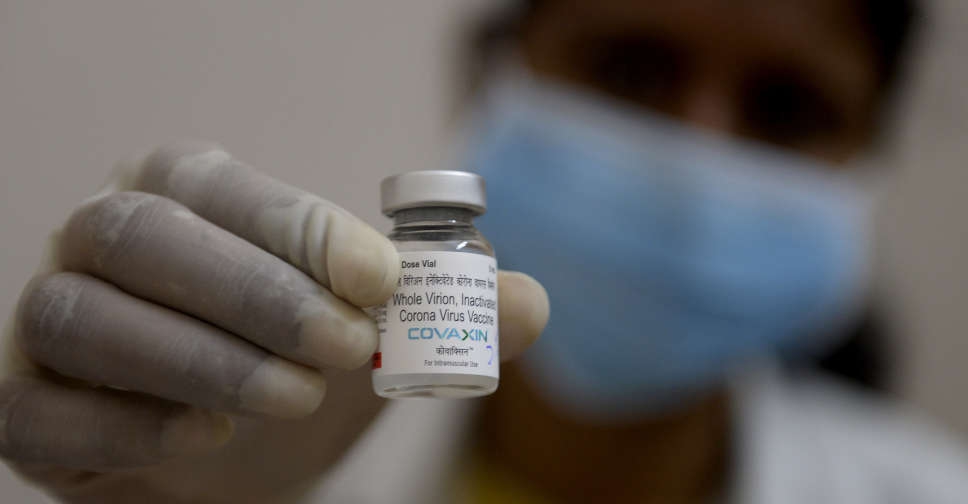
A World Health Organisation (WHO) technical advisory group was reviewing data on India's Covaxin shot against COVID-19 on Tuesday with a decision on its emergency use listing likely soon, a spokesperson said.
"If all is in place and all goes well and if the committee is satisfied, we would expect a recommendation within the next 24 hours or so," Margaret Harris told journalists at a UN press briefing.
Millions of Indians have taken the shot produced by Bharat Biotech but many have been unable to travel pending the WHO approval.




 Iran denies talks with US after Trump postpones strikes on power grid
Iran denies talks with US after Trump postpones strikes on power grid
 Kim Jong Un says North Korea’s nuclear status is irreversible, threatens South
Kim Jong Un says North Korea’s nuclear status is irreversible, threatens South
 At least 34 killed in Colombia military plane crash, governor says
At least 34 killed in Colombia military plane crash, governor says
 Ukraine's Zelenskyy says Russia preparing new massive attack
Ukraine's Zelenskyy says Russia preparing new massive attack
 Rockets launched from Iraq’s Mosul towards US base in Syria
Rockets launched from Iraq’s Mosul towards US base in Syria




